

Mutations in Parkin, essential for degradation of damaged mitochondria, cause early-onset PD. Objective: To establish a workflow for mitochondrial DNA (mtDNA) CpG methylation using Nanopore whole-genome sequencing and perform first pilot experiments on affected Parkin biallelic mutation carriers (Parkin-PD) and healthy controls.Background: Mitochondria, including mtDNA, are established key players in Parkinson's disease (PD) pathogenesis.

By leveraging seven human cell lines that are designated as publicly available reference materials, these data can be used as a baseline to advance epigenomics research. The data provided herein can guide the use of these DNA reference materials in epigenomics research, as well as provide best practices for experimental design in future studies. After rigorous quality assessment and comparison to Illumina EPIC methylation microarrays and testing on a range of algorithms (Bismark, BitmapperBS, bwa-meth, and BitMapperBS), we find overall high concordance between assays, but also differences in efficiency of read mapping, CpG capture, coverage, and platform performance, and variable performance across 26 microarray normalization algorithms. Here, we present a multi-platform assessment and cross-validated resource for epigenetics research from the FDA’s Epigenomics Quality Control Group.Įach sample is processed in multiple replicates by three whole-genome bisulfite sequencing (WGBS) protocols (TruSeq DNA methylation, Accel-NGS MethylSeq, and SPLAT), oxidative bisulfite sequencing (TrueMethyl), enzymatic deamination method (EMSeq), targeted methylation sequencing (Illumina Methyl Capture EPIC), single-molecule long-read nanopore sequencing from Oxford Nanopore Technologies, and 850k Illumina methylation arrays. However, the wide variety of approaches available to interrogate these modifications has created a need for harmonized materials, methods, and rigorous benchmarking to improve genome-wide methylome sequencing applications in clinical and basic research. By optimizing the standard ONS library preparation, we achieved selective enrichment of native mtDNA and accurate single nucleotide variant and CpG methylation calling, thus overcoming previous limitations.Ĭytosine modifications in DNA such as 5-methylcytosine (5mC) underlie a broad range of developmental processes, maintain cellular lineage specification, and can define or stratify types of cancer and other diseases. Here, we propose an alternative protocol to quantify methyl-CpGs in mtDNA, at single-molecule level, using Oxford Nanopore Sequencing (ONS). Lastly, calling mtDNA alleles with varying proportions (heteroplasmy) is complicated by the C-to-T conversion introduced by WGBS on unmethylated CpGs.
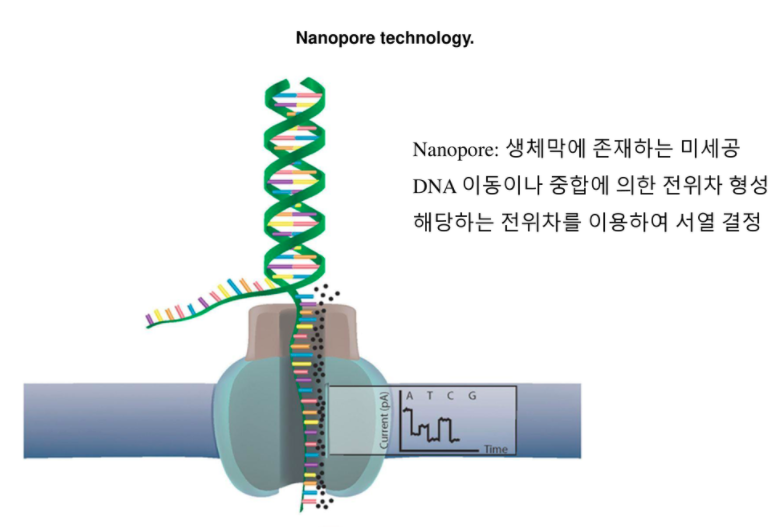
Also, short-read sequencing can result in a co-amplification of nuclear sequences originating from ancestral mtDNA with a high nucleotide similarity. Concerns include mtDNA strand asymmetry rendering the C-rich light strand disproportionately vulnerable the chemical modifications introduced with WGBS. In conclusion, our protocol enables the reliable analysis of epigenetic modifications of mtDNA at single-molecule level at single base resolution, with potential applications beyond methylation.MotivationAlthough whole genome bisulfite sequencing (WGBS) is the gold-standard approach to determine base-level CpG methylation in the nuclear genome, emerging technical issues raise questions about its reliability for evaluating mitochondrial DNA (mtDNA) methylation. Methylation calling revealed negligible mtDNA methylation levels in multiple human primary and cancer cell lines. Variant calling analysis against Illumina deep re-sequencing showed that all expected mtDNA variants can be reliably identified. Our approach circumvents mtDNA-specific confounders, while enriching for native full-length molecules over nuclear DNA. Here, we validate the technical concerns with WGBS, and then develop and assess the accuracy of a protocol for variant-specific methylation identification using long-read Oxford Nanopore Sequencing. Methylation of mitochondrial DNA (mtDNA) has been studied using whole genome bisulfite sequencing (WGBS), but recent evidence has uncovered major technical issues which introduce a potential bias during methylation quantification. SummaryMethylation on CpG residues is one of the most important epigenetic modifications of nuclear DNA, regulating gene expression.

